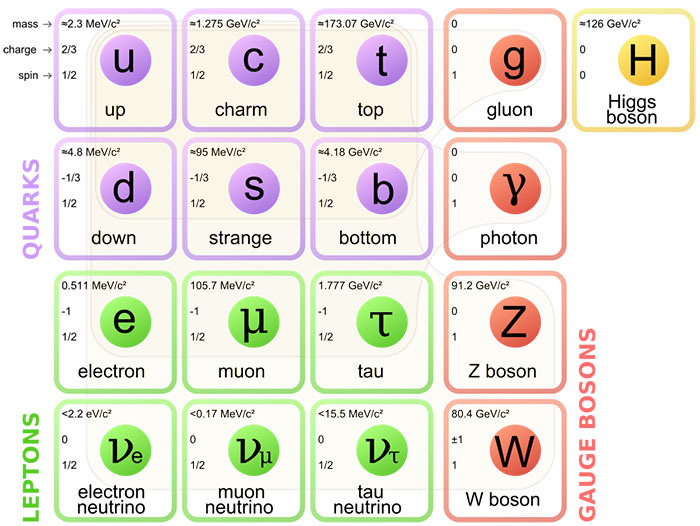
P3: Fermions
The other side of the Standard Model; The particles that make up the surrounding matter: Fermions. Let's explore the different kinds of Fermions.
1) Shared Characteristics of Fermions
On the Standard Model above, our Fermions consist of both our Quarks and Leptons. They are characterized by their Spin of 1/2, with Fermions sharing this Half-Integer Spin, contrasting the whole integer spin of Bosons (0/1).
As shown above, the Fermions are separated into 2 Elementary Classes: Quarks and Leptons.
2) Quarks
Quarks are the building blocks of most particles around us. As an example, Nucleons (Protons/Neutrons) are made up of 3 Quarks:
The Proton consists of 2 Up and 1 Down Quark: Net Charge of +1
The Neutron consists of 2 Down and 1 Up Quark: Net Charge of 0
Unlike other particles, Quarks cannot exist independently, and instead exist in some combination, with the combination of quarks resulting in particles of different characteristics. Particles made up of Quarks are known as Hadrons.
The Quarks can also be changed - through radioactive decay - which can cause one particle to change into another type of particle.
This is an entirely separate subject, but a quick example would be Beta Minus Decay, where a Neutron changes into a Proton, while ejecting an Electron and Antineutrino (other small particles) and emitting a W Boson.
The Quark configuration changes from udd (Up-Down-Down for Neutrons) to uud (Up-Up-Down for Protons)
3) Leptons
Unlike Quarks, Leptons can exist independently as Lone Particles, with each Lepton having its unique characteristics.
Electrons: These are the most commonly encountered Leptons, and are considered to be stable, with a relative charge of -1.
Muons: These are produced in the atmosphere from Cosmic Rays showering the Atmosphere, and though they have the same relative charge as Electrons, they’re much heavier, and decay very quickly, with a life of 2.2 microseconds.
Tau: These are the highest mass of all the Leptons, and are primarily found at the core of stars, or produced in Particle Accelerators. They share the same relative charge as Electrons, and decay even faster than Muons at 0.29 Picoseconds.
Muons and Tau Particles can both just be thought of as heavier electrons, for simplicity's sake, since aside from mass, many of their Fundamental properties are identical to those of Electrons. With this increase in mass, they’re also more unstable, which explains their short lifespans.
As for the other 3 - the Neutrinos - they’re more complex. Neutrinos are very light particles that only interact via 2 of the 4 Fundamental Forces: Gravity and the Weak Nuclear Force. I won’t discuss any further distinctions between them, since they’re similar in idea to the other 3 Leptons:
The Electron-Neutrino has the lowest mass; otherwise, these 3 Neutrinos share the same Fundamental Properties.
Summary:
There are two smaller classes of Fermions.
Quarks cannot exist independently, and exist as combinations, with the resultant particles being known as Hadrons, which can have different properties depending on the kind of Quarks.
Leptons can exist independently, or be extracted, with the non-neutrino leptons all sharing similar traits to electrons, except for mass and stability.
And with that, you should now have a fundamental understanding of the different kinds of Fermions. Note that there is more complex physics governing each type of Fermion, and there are specific interactions, but I’ll address them in the future.
4) Differences between Bosons and Fermions
The first difference is that Fermions have a half-integer Spin, whilst Bosons have a whole-integer spin value (0 or 1)
The other difference relates to a concept called the Pauli-Exclusion Principle, which I’ve covered before. The simplified form is that, for a given Quantum State (where particles have a specific position, and occupy discrete, specific energy levels), identical Bosons can exist in the same energy state due to them having Symmetric Wave Functions. By contrast, identical Fermions can’t exist in the same energy state, or position in space, due to them having Asymmetric Wave Functions.
This is why identical electrons can’t occupy the same Orbital, and so the 2 electrons that typically occupy an Electron Orbital must have some difference.
Sources for Further Reading:
http://https//www.youtube.com/watch?v=_1S6KfMzOH8
http://https//www.youtube.com/watch?v=skFU7pmBOys